In children with cystic fibrosis (CF) or other forms of malabsorption, vitamin D doesn’t merely “supplement” a diet—it performs a quiet choreography. It escorts calcium into the skeleton, instructs the immune system in measured restraint, and helps muscles respond with less fatigue and more resilience. Yet the path of vitamin D is rarely straightforward. Malabsorption can turn a seemingly simple nutrient into a dwindling traveler, slipping through digestive “cracks” before it can do its work. This is why vitamin D strategy becomes less like a routine pill and more like an ongoing, detective-like practice: measure, interpret, and tailor.
The gut as a gatekeeper: why malabsorption changes everything
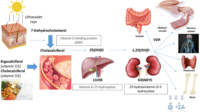
Vitamin D is fat-soluble, so it prefers to travel with lipids. In malabsorption, especially in CF where pancreatic enzymes may be insufficient, fats may not be digested or absorbed efficiently. When absorption falters, vitamin D can become an underfed signal—present in the bloodstream only in fits and starts. The result can be lower serum 25-hydroxyvitamin D, altered calcium balance, and compensatory hormonal shifts that quietly siphon strength from bones.
Think of the digestive system as a museum with a narrow door. Vitamin D is a valuable artifact, but if the museum’s ticketing system—enzymes, bile flow, and micelle formation—is inconsistent, fewer artifacts get through each day. The child may still eat, but the body’s “transit authority” cannot reliably deliver vitamin D to where it’s needed most.
Why cystic fibrosis requires a more vigilant vitamin D lens
CF is not only a lung story; it’s a whole-body story with nutrition at the center. Many children with CF have exocrine pancreatic insufficiency, and therefore rely on pancreatic enzyme replacement therapy (PERT). When PERT dosing is optimal and meals are well-timed, absorption can improve. Still, even with careful management, vitamin D needs may remain higher than average because losses can occur through multiple pathways—insufficient intake, altered absorption, increased metabolic demands, and differences in activity levels.
Vitamin D in CF is often treated like a “diagnostic compass.” When levels are low, bone health, muscle function, and overall well-being can be affected. When levels normalize, children frequently feel stronger, more stable, and sometimes experience fewer musculoskeletal complaints. The compelling part is that the strategy can be both data-driven and emotionally reassuring—parents gain a tangible target, and clinicians gain a modifiable lever.
Blood tests: the map before the journey
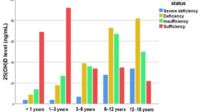
The most informative marker is typically serum 25-hydroxyvitamin D. It reflects vitamin D status more reliably than shorter-acting measures. Testing also helps clinicians interpret whether symptoms and lab results align, particularly when there’s concern for bone mineral density issues or nutritional strain.
Clinicians may also check calcium, phosphorus, alkaline phosphatase, and parathyroid hormone. These values act like “supporting characters” that confirm the plot. For example, elevated parathyroid hormone can suggest that calcium balance is struggling, which may indicate a functional vitamin D deficit even if the picture is not perfectly linear.
Targeting bone health: beyond deficiency, into prevention
Vitamin D supports mineralization—the process where bones harden into strength instead of staying soft and vulnerable. In children, inadequate vitamin D can increase the risk of rickets, but CF-related risks often extend beyond frank deficiency. Even suboptimal levels may contribute to reduced bone density, delayed mineralization, and higher fracture susceptibility later in life.
Malabsorption turns bone health into a moving target. A child may grow quickly, and growth itself is a high-demand season for calcium and vitamin D. Picture a construction crew building a city at high speed. If vitamin D supplies are unreliable, the mortar can be thin, and the city may look complete—until stress reveals the seams.
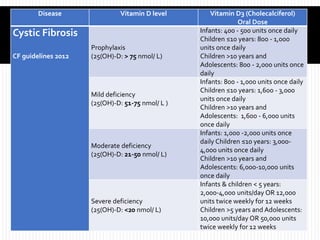
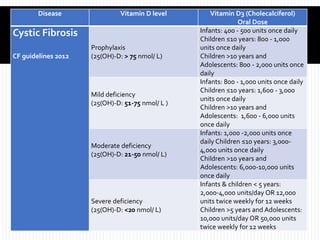
Dosing principles: individualized, meal-aware, and clinically monitored
Vitamin D dosing is rarely one-size-fits-all. Age, baseline serum levels, dietary intake, severity of malabsorption, adherence to PERT, and coexisting conditions all matter. Some children require higher daily doses; others may use intermittent supplementation regimens, especially when adherence is challenging.
Because vitamin D is fat-soluble, dosing strategies often emphasize taking supplements with meals that contain fat. In CF, aligning vitamin D intake with effective PERT timing can be particularly meaningful. If PERT is delayed or underdosed, vitamin D may arrive in the bloodstream but fail to achieve its full destination.
After initiating or adjusting therapy, follow-up testing is typically used to confirm response. Short intervals may be chosen to ensure levels rise appropriately; longer intervals may be used when stability is achieved. Monitoring reduces the risk of overshooting—because too much vitamin D can also disrupt calcium regulation.
Vitamin D forms: selecting the right vehicle for the nutrient
Vitamin D can be supplied as vitamin D2 (ergocalciferol) or vitamin D3 (cholecalciferol). The choice may vary by clinical preference, availability, and regional practice. Regardless of form, the goal is consistent and adequate absorption and a measurable improvement in serum 25-hydroxyvitamin D.
In a malabsorptive environment, consistency is the true companion. A child who takes a supplement intermittently may never achieve a stable vitamin D “baseline,” similar to a lighthouse that flashes but never holds steady illumination. Clinicians therefore weigh not only the dose but also the child’s practical ability to take it—especially during busy school schedules or periods of illness.
Synergy with calcium, enzymes, and overall nutrition
Vitamin D does not work alone. Calcium status, dietary protein adequacy, and overall energy intake influence outcomes. In CF, PERT is often foundational; without adequate enzyme coverage for meals, vitamin D and calcium absorption may remain compromised.
This is why nutrition planning is frequently multidisciplinary. Dietitians may adjust caloric goals, refine meal composition, and help families build routines that make enzyme use practical. When nutrition becomes cohesive, vitamin D has a better chance to perform its role as a conductor—coordinating calcium availability and supporting bone remodeling.
Sometimes children also require attention to magnesium or other micronutrients, depending on lab results and clinical context. The narrative is consistent: malabsorption isn’t a single broken gear; it’s a system, and the solution is likewise systemic.
Growth, symptoms, and when to act quickly
Vitamin D issues can be silent. A child may not complain of pain until mineralization has already been affected. Still, clues can appear: bone pain, muscle weakness, delayed growth, fatigue, unusual gait, or concerns about fractures. In CF, frequent respiratory infections and overall nutritional strain can also coincide with metabolic imbalances.
When symptoms and labs suggest deficiency or insufficiency, timely adjustment matters. The most intriguing aspect of vitamin D therapy is that it can sometimes turn the tempo back toward recovery—supporting strength and function while also protecting the skeleton’s future.
Safety and the balancing act: preventing both deficiency and excess
Healthy supplementation is a balancing act. Clinicians aim for enough vitamin D to normalize serum levels and support bone health, while avoiding excessive intake that may increase calcium levels and cause symptoms such as nausea, constipation, or lethargy.
Regular monitoring and clear dosing instructions help families feel confident. In the story of vitamin D, safety is not an afterthought—it is the plot’s steady anchor. With careful follow-up, supplementation becomes a controlled intervention rather than a gamble.
Building a sustainable routine: adherence as a therapeutic tool
Compliance is not merely a behavioral challenge; it is a medical strategy. Short-term schedules fail when life is chaotic, so vitamin D plans often need to fit into real routines: school mornings, meal times, and enzyme dosing patterns. Some families find success with consistent timing and reminders; others benefit from simplified regimens.
It helps to frame vitamin D as a protective ritual, not a punishment. Over time, families can transform supplementation into a predictable step—like buckling a seatbelt before the car moves. The child doesn’t need to fear it; the family simply integrates it into the day’s rhythm.
Looking ahead: long-term monitoring and lifelong skeletal stewardship
For children with CF or ongoing malabsorption, vitamin D management is often an ongoing chapter. Levels may fluctuate with illness, changes in appetite, hospitalizations, or adjustments to enzyme and nutrition plans. Bone health can also evolve with age, puberty, and activity levels.
Long-term follow-up may include periodic lab reassessment and, when indicated, bone density evaluation. The purpose is to keep the skeleton—and the child’s energy and confidence—on a sturdy trajectory.
Vitamin D for children with CF or malabsorption is best understood as an orchestration. When measurement guides dosing, meals and enzymes align, and safety is monitored, vitamin D can shift from a missing note to a sustaining theme. The goal isn’t simply to raise a number on a lab report. It’s to help a growing child build durable strength—brick by brick—through a nutrient pathway that was once unreliable.