There is a particular kind of medical curiosity that arrives quietly—at first as a pattern in observational studies, later as a biological plausibility that refuses to be dismissed. Polycystic Ovary Syndrome (PCOS) is notorious for its variability: irregular ovulation, androgen excess, metabolic turbulence, and the unsettling complexity that makes every patient’s story feel distinct. And then, like a subtle reframing of the landscape, vitamin D enters the conversation. Not as a miracle solvent, not as a single lever that “fixes” PCOS, but as a potential modulator—an immunometabolic signal with the capacity to nudge reproductive endocrinology in a different direction.
Vitamin D: More Than a Sunlight Myth
Vitamin D is often introduced as a bone-and-skin nutrient, yet its actual influence is broader and more theatrical. In the body, it behaves like a hormone precursor, interacting with nuclear receptors that regulate gene transcription. Those receptors appear in tissues relevant to PCOS—ovaries, adipose tissue, pancreatic islets, immune cells, and the vascular system.
This matters because PCOS is not merely reproductive; it is also inflammatory and metabolic. Vitamin D’s involvement in insulin sensitivity, inflammatory signaling, and cellular differentiation suggests it could shape the endocrine “tone” in which ovarian function unfolds. The shift in perspective is subtle but profound: instead of thinking of vitamin D as a deficiency to correct, consider it as a regulatory language that may be missing fluency in some people with PCOS.
PCOS at a Glance: Why Multifactorial Means Multifield
PCOS typically emerges from a convergence of mechanisms: ovulatory dysfunction, hyperandrogenism, and metabolic irregularity—often accompanied by chronic low-grade inflammation. Many patients also experience dyslipidemia, impaired glucose handling, and weight-related challenges, though the distribution of symptoms varies. Some carry insulin resistance as a dominant theme; others display reproductive symptoms with less overt metabolic disruption.
This heterogeneity is precisely why vitamin D is compelling. A compound that participates in multiple pathways—immune modulation, insulin signaling, steroidogenesis—can plausibly affect more than one clinical domain. Vitamin D doesn’t have to “solve” PCOS in one stroke; it may instead contribute to a more stable endocrine environment.
Evidence Connections: What the Studies Suggest (and What They Don’t)
The research landscape around vitamin D and PCOS is dynamic—promising, sometimes inconsistent, and always informative. Many studies report that people with PCOS experience a higher prevalence of vitamin D insufficiency or deficiency. That observation creates an intuitive hypothesis: low vitamin D might be part of the PCOS puzzle.
However, causality is harder to pin down. PCOS can influence behaviors and physiology that affect vitamin D status—for example, body fat distribution, lifestyle patterns, and metabolic factors that alter vitamin D handling. The chicken-and-egg dilemma lingers. Yet even without perfect causal certainty, patterns can be clinically useful: if deficiency is common, correcting it may provide additional benefit, especially in a body already juggling inflammation and insulin dysregulation.
Beyond prevalence, trials exploring vitamin D supplementation have investigated outcomes such as androgen levels, insulin sensitivity markers, menstrual regularity, and measures of ovulatory function. Results have varied, but the most interesting theme is directionality: many trials show improvements in metabolic or hormonal parameters, even if not every endpoint responds.
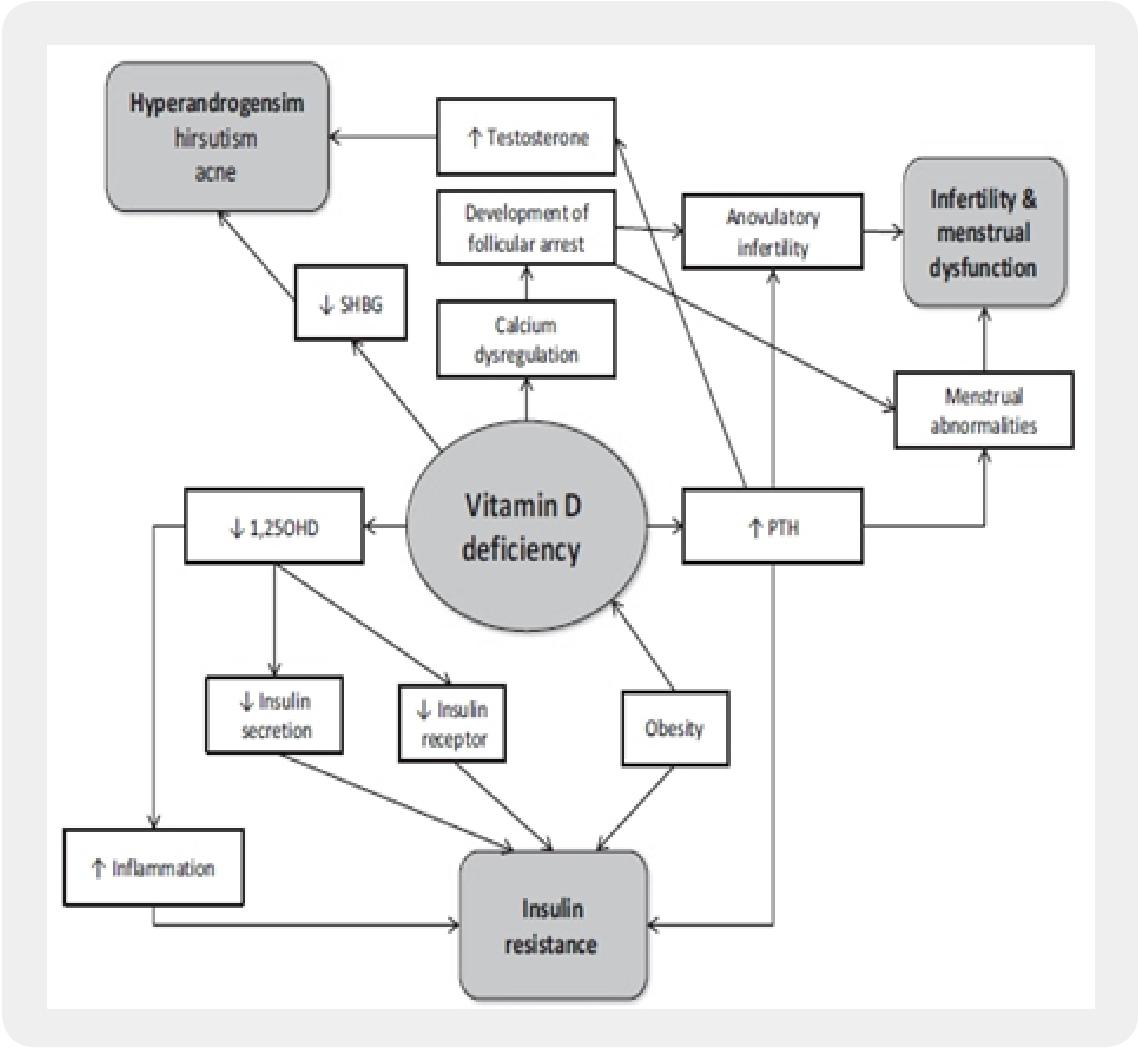
Immunometabolic Pathways: The Hidden Bridge
One of the most fascinating links between vitamin D and PCOS is immunometabolism—the interweaving of immune activity with metabolic control. PCOS is often characterized by an inflammatory milieu that can impair insulin signaling and disturb ovarian steroidogenesis.
Vitamin D may help recalibrate immune signaling by influencing cytokine production and immune cell behavior. This could, in turn, improve insulin sensitivity. When insulin resistance becomes less aggressive, ovarian theca cells may receive a different metabolic “message,” potentially reducing hyperandrogenism. The domino effect is not guaranteed, but the logic is coherent.
In other words, vitamin D might not act like a hammer. It may act like a dimmer switch—altering the intensity of signals that drive PCOS physiology.
Insulin Resistance and Androgens: A Feedback Loop Worth Watching
Insulin is not just a metabolic hormone; it also interacts with ovarian androgen production. Elevated insulin levels can stimulate androgen synthesis and influence ovarian microenvironments. This creates a feedback loop where metabolic disruption supports endocrine disruption.
Vitamin D’s potential to improve insulin sensitivity could therefore affect androgen levels indirectly. Some research suggests that supplementation may lower circulating markers associated with androgen excess or improve related clinical features. Even modest hormonal shifts can matter, because androgen and insulin dynamics influence follicular maturation and ovulatory scheduling.
The curiosity here is practical: if the loop can be softened, ovarian function might become more regular. That doesn’t imply immediate transformation, but it points toward a plausible mechanism for gradual improvement.
Menstrual Regularity and Ovulatory Function: The Reproductive Payoff
For many people, the most visible impact of PCOS is menstrual irregularity—cycles that arrive unpredictably, ovulation that is inconsistent, and the emotional weight of uncertainty. Trials that examine reproductive endpoints aim to determine whether vitamin D supplementation can shift ovulatory function or support more regular bleeding patterns.
Outcomes vary, yet the concept remains compelling: by influencing insulin sensitivity and inflammatory tone, vitamin D could help restore ovarian signaling to a more orderly rhythm. Short-term supplementation might not immediately “rebuild” reproductive cycles, but longer interventions and careful monitoring could reveal benefits in certain subgroups.
This subgroup principle is key. PCOS is not one monolith; vitamin D may be more impactful where deficiency is profound and metabolic disturbance is active.
Body Weight, Adipose Biology, and Vitamin D Sequestration
Adipose tissue is not passive storage—it is an endocrine organ. It also has a complicated relationship with vitamin D. In individuals with higher body fat, vitamin D can become sequestered in adipose compartments, lowering bioavailability in circulation. Meanwhile, adipose inflammation can amplify metabolic strain.
This is where vitamin D becomes less of a single-variable story and more of a systems story. Correcting deficiency may work differently depending on body composition, baseline vitamin D status, and lifestyle patterns. Weight changes—whether through diet, exercise, or medical therapies—can also influence vitamin D dynamics, complicating interpretations of supplementation outcomes.
Yet the combined possibility remains interesting: improvements in metabolic health and reductions in inflammatory signaling could synergize with vitamin D repletion.
Clinical Safety and Practical Supplementation Considerations
Vitamin D supplementation is widely available, but “available” is not the same as “automatically appropriate.” The safest and most effective approach usually begins with assessment. Measuring baseline vitamin D status helps avoid guessing, because the required dose depends on whether someone is insufficient, deficient, or simply hovering near a borderline range.
Dose regimens differ across clinical guidance, and the goal typically involves repletion followed by maintenance. Equally important is monitoring. Excessive vitamin D intake can cause hypercalcemia, so a disciplined approach matters—especially for individuals with kidney disease, granulomatous disorders, or other conditions that alter calcium handling.
In a PCOS context, supplementation should be considered alongside broader management strategies, not as a standalone replacement for established care.
Who Might Benefit Most? The Subgroup Hypothesis
Rather than asking whether vitamin D “works” universally, a more useful question is: in whom might vitamin D yield the most noticeable benefit? Several plausible factors include baseline deficiency severity, insulin resistance intensity, inflammatory biomarkers, body composition, and adherence to supplementation.
Age and coexisting conditions may also shape response. Some individuals could experience improvements in insulin sensitivity without large hormonal shifts, while others might notice clearer changes in cycle regularity. The variability does not necessarily negate vitamin D’s role; it may simply reflect the heterogeneity of PCOS itself.
Future research that stratifies participants—by deficiency status, metabolic phenotype, and inflammatory state—could sharpen the clinical forecast.
Integrating Vitamin D Into a Broader PCOS Strategy
PCOS management is most effective when it is multidimensional. Lifestyle interventions that target insulin sensitivity—such as dietary refinement and resistance or aerobic exercise—remain foundational. When fertility is a goal, ovulation induction strategies may be necessary. For androgen-related symptoms, targeted treatments may help.
In that larger ecosystem, vitamin D may act as an adjunct: a nutritional signal that can support metabolic stability and immune regulation. It is rarely the sole intervention, but it can be a meaningful piece of the puzzle, particularly for those with confirmed deficiency or insufficient levels.
Future Directions: From Correlation to Precision
The next phase of exploration is precision-oriented. Researchers increasingly recognize that PCOS is not a single disease mechanism; it is a syndrome with multiple biological “faces.” Clarifying which pathways vitamin D most strongly influences—immune signaling, insulin sensitivity, androgen regulation, follicular maturation—may determine where supplementation has the best odds of producing tangible clinical change.
Longer trials, better phenotyping, and standardized dosing strategies could reduce inconsistency and reveal patterns that current studies only hint at. The ultimate aspiration is not maximal vitamin D exposure; it is optimal endocrine coordination.
A Curiosity-Driven Conclusion: Reframing What Help Can Look Like
Vitamin D and PCOS sit at an intriguing intersection of endocrinology, metabolism, and immune biology. The evidence does not demand belief in a single, sweeping cure. Instead, it invites a different mindset: treat vitamin D insufficiency seriously, regard supplementation as a potential modulator, and evaluate response within the broader PCOS context.
For some, correcting deficiency may unlock incremental improvements—better metabolic resilience, softened inflammatory tone, and possibly more regular ovarian signaling. For others, vitamin D may play a smaller role, but still offers a rational, low-cost axis to consider.
Curiosity is not merely academic here. It can shape better conversations, smarter screening, and more personalized management—turning a nutrient once dismissed as “sunlight-related” into a potential ally in the complex choreography of PCOS.








