Every few months, a familiar question reappears in conversations about women’s health: “Can vitamin D really influence breast cancer risk?” The observation is understandable—vitamin D seems like such a modest nutrient, often tied to sunny days and routine bloodwork. Yet the fascination runs deeper. Vitamin D doesn’t behave like a simple vitamin; it functions more like a molecular interpreter, translating sunlight-derived signals into pathways that govern cell behavior. And when those pathways wobble, biology may look for another story to tell—sometimes one written in the language of cancer.
Why breast tissue pays attention to vitamin D
Breast tissue is not a passive bystander. It contains receptors that respond to vitamin D, allowing the body to influence gene expression, regulate cell growth, and modulate inflammatory signaling. This is where the common observation begins to feel incomplete. We often treat cancer risk as a single-variable equation—diet here, genetics there, screenings somewhere else. But risk is usually a tapestry of influences stitched together over time.
Vitamin D’s role can be imagined as a tuning mechanism. When levels are adequate, cells may receive clearer instructions about differentiation (becoming the “right kind” of cell), apoptosis (the self-cleaning of damaged cells), and immune surveillance. When levels are chronically low, those instructions can become muffled. It’s not that vitamin D alone decides fate; rather, it may shape the stage on which other risk factors act.
The “low vitamin D” pattern and the instinct to generalize
Many women naturally wonder whether a single marker—25-hydroxyvitamin D—can explain why some people experience higher incidence rates. The temptation is to generalize: if vitamin D is low, risk is higher. The intrigue deepens because low vitamin D is common. People spend less time outdoors, cultures favor indoor living, and skin pigmentation and latitude can influence vitamin D synthesis. Add darker seasons, indoor clothing, and screen-heavy routines, and deficiency can become a quiet background rhythm rather than an occasional event.
But here is the important nuance: low vitamin D is both a possible contributor and a possible surrogate. It may reflect reduced sun exposure, which itself can correlate with vitamin D status, but also with lifestyle patterns—activity levels, body composition, and overall metabolic health. In other words, vitamin D might be part of the causal chain, or it might be an indicator of broader physiologic conditions. Often, real life is messier than laboratory isolation.
Mechanisms: how vitamin D may help shift cancer risk biology
Vitamin D engages multiple biological circuits relevant to cancer development. One mechanism involves controlling cell proliferation—how quickly cells divide. Another involves differentiation, steering cells away from a “chaotic growth” trajectory. Vitamin D also participates in the inflammatory orchestra. Chronic inflammation can behave like static in the immune system’s communication lines, making it harder to notice and eliminate abnormal cells.
There’s also the matter of hormone signaling. Breast cancer is frequently influenced by estrogen and related pathways, and vitamin D may interact with hormonal regulation indirectly through immune function and cellular growth control. The deeper fascination is that vitamin D’s effects are not limited to one pathway; it can resemble a multi-tool rather than a single-purpose instrument.
These mechanisms help explain why vitamin D is discussed not only as “prevention” but as risk-modifying support—potentially influencing how the body handles damaged cells, oxidative stress, and immune competence.
Vitamin D and the immune system: surveillance at the microscopic level
Immune surveillance is like a night watch. It doesn’t guarantee perfection, but it can reduce the chance that abnormal cells survive long enough to establish a foothold. Vitamin D supports components of both innate and adaptive immunity. It may help immune cells adopt a more balanced posture—capable of attacking threats without tipping into chronic, dysregulated inflammation.
When vitamin D status is insufficient, immune signaling may become less coordinated. Abnormal cells might escape detection more easily, and inflammation-related microenvironments may become more permissive. This is one reason many researchers view vitamin D status as a “context variable,” altering how risk factors play out over years.
What “risk reduction” really means in practice
Risk reduction is rarely a dramatic on-off switch. It’s more like nudging a long-running process toward a safer direction. For breast cancer risk, this can include multiple layers: maintaining cellular regulation, supporting immune activity, and addressing deficiency that might correlate with other health vulnerabilities.
Importantly, vitamin D should not be framed as a replacement for established prevention strategies. Screening, genetic risk assessment, lifestyle measures, and appropriate medical guidance remain central. Think of vitamin D as one thread—valuable, but best integrated into a broader fabric of care.
How to think about dose: supplements, sunlight, and blood levels
Because vitamin D status varies, the most sensible starting point is measurement. Many clinicians use 25-hydroxyvitamin D as the circulating snapshot. From there, the goal is often to support adequate range for skeletal health and potential extra-skeletal signaling.
Sunlight can help, but it is inconsistent—season, latitude, cloud cover, sunscreen habits, and time outdoors can all influence production. Supplements can create steadier intake, yet they still require personalization. Body weight, absorption efficiency, baseline levels, and concurrent medications can all modify needs.
A common misconception is that more is always better. Vitamin D has a physiological ceiling, and excess can be harmful. The more strategic approach is careful titration, monitored by testing and guided by healthcare professionals.
Breast cancer is not one disease: heterogeneity matters
“Breast cancer” is an umbrella word. There are multiple subtypes with different biological behaviors—some more hormone-driven, others with distinct cellular features. Vitamin D’s influence may vary depending on subtype, stage, and the surrounding microenvironment.
This is part of why some studies show associations while others yield mixed results. When biology is heterogeneous, the “average effect” can blur. Still, the broader concept remains compelling: vitamin D participates in pathways that are relevant to tumor development and progression.
Body composition, metabolism, and the vitamin D feedback loop
Another hidden layer is body composition. Vitamin D is fat-soluble, and higher adiposity can be associated with lower circulating levels. Metabolic health—insulin sensitivity, chronic inflammation, and lipid-related signaling—also intersects with cancer risk.
In that sense, low vitamin D may travel alongside metabolic changes rather than act alone. Yet the feedback loop is not hopeless. Addressing vitamin D insufficiency, supporting healthy activity, and improving metabolic markers can work together, potentially creating a more favorable internal environment.
Life stage and consistency: the “years matter” perspective
Breast tissue experiences decades of biological fluctuation—hormonal rhythms, reproductive factors, aging-related immune changes, and repeated exposure to environmental stressors. Vitamin D status can shift during adulthood due to lifestyle changes, weight variation, and seasonal patterns.
This is why consistency matters. Correcting deficiency once may be helpful, but sustained adequacy often matters more. Think long-range: not “one good month,” but “a reliable baseline over time.” The deeper fascination here is that biology is patient; it keeps records through chronic exposure, not just acute events.
What helps alongside vitamin D: a risk-reduction ecosystem
Vitamin D works best when embedded in a coherent strategy. Physical activity supports immune function and may influence hormonal regulation. A nutrient-dense diet can reduce oxidative stress and inflammation. Adequate protein and micronutrients support healthy tissue repair. Limiting alcohol and avoiding smoking further reduce risk.
Even stress management and sleep quality—often treated as peripheral—can influence inflammation and endocrine signals. This is not mysticism; it’s physiology. The body is an interconnected system, and breast cancer risk is shaped by that connectivity.
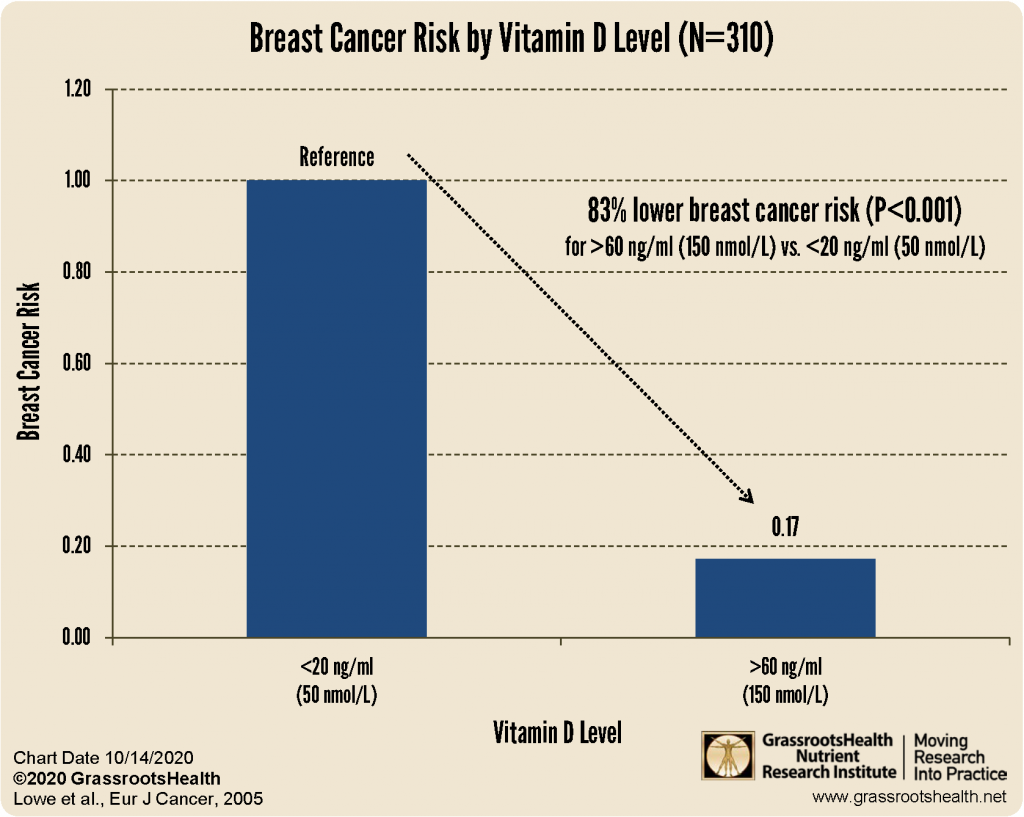
Visual reassurance and the decision point
When women see charts or infographics comparing vitamin D status and breast cancer outcomes, it can feel both hopeful and overwhelming. Charts summarize complexity into a glanceable pattern, and the real decision begins afterward: testing, conversation with a clinician, and a practical plan.
Below is an example-style visual that reflects how the topic is often presented to the public—helpful as a starting cue, but best followed by evidence-informed action.

If a woman’s level is low, she may ask not just “How do I supplement?” but “How do I integrate vitamin D into a long-term health architecture?” When vitamin D is approached as part of a broader ecosystem—sleep, movement, nutrition, screening, and personalized medical guidance—its potential becomes more grounded and less speculative.
A final, grounded takeaway
Vitamin D and breast cancer risk reduction are compelling because the nutrient is biologically plausible. It influences cell regulation, immune function, and inflammatory signaling—processes intimately involved in cancer development. Still, risk reduction is not guaranteed by one measure, and associations do not equal destiny.
The most constructive path is the middle one: measure vitamin D status, correct insufficiency safely and consistently when appropriate, and maintain the other well-established protective behaviors. In that balanced approach, vitamin D becomes what it always was—a signal from sunlight and diet, translated into cellular guidance, offered to support a healthier trajectory.