Inflammation is less a single event than a chronic storyline running through the body—quietly at first, then louder when the immune system starts behaving like a miscalibrated alarm. In the search for calmer physiology, two names keep surfacing: vitamin D and GLP-1–related therapies. At a glance, they belong to different worlds—bone biology on one side, metabolic signaling on the other. Yet increasing evidence suggests they may intersect in a meaningful way, influencing inflammatory pathways, immune tone, and tissue repair. The link is not merely a headline; it’s a network of molecular conversations that can shape how skin, joints, and the gut respond to stress.
Vitamin D as an Inflammation Modulator, Not Just a “Vitamin”
Vitamin D is often discussed as if it were a simple nutrient, the kind you either have or don’t. But biologically, it behaves more like a hormone-shaped coordinator. Once converted into its active form, it can influence gene transcription in immune cells—think of T cells, macrophages, and dendritic cells—nudging them toward a less inflammatory phenotype.
One important mechanism involves dampening pro-inflammatory cytokines—small chemical “messengers” that amplify immune responses. When vitamin D levels are adequate, these signals may be less incessant. Meanwhile, it can support anti-inflammatory mediators and promote immune regulation. The result is not “shutting down” immunity. It’s more like turning down the volume while keeping the band ready to play.
Readers often find it useful to understand that vitamin D status can be affected by season, latitude, skin pigmentation, diet, and sun exposure. Low levels are common, particularly in people with darker skin or limited daylight. That deficiency state can set the stage for inflammatory signaling to become more pronounced.
GLP-1 Signaling: A Metabolic Pathway with Immune Consequences
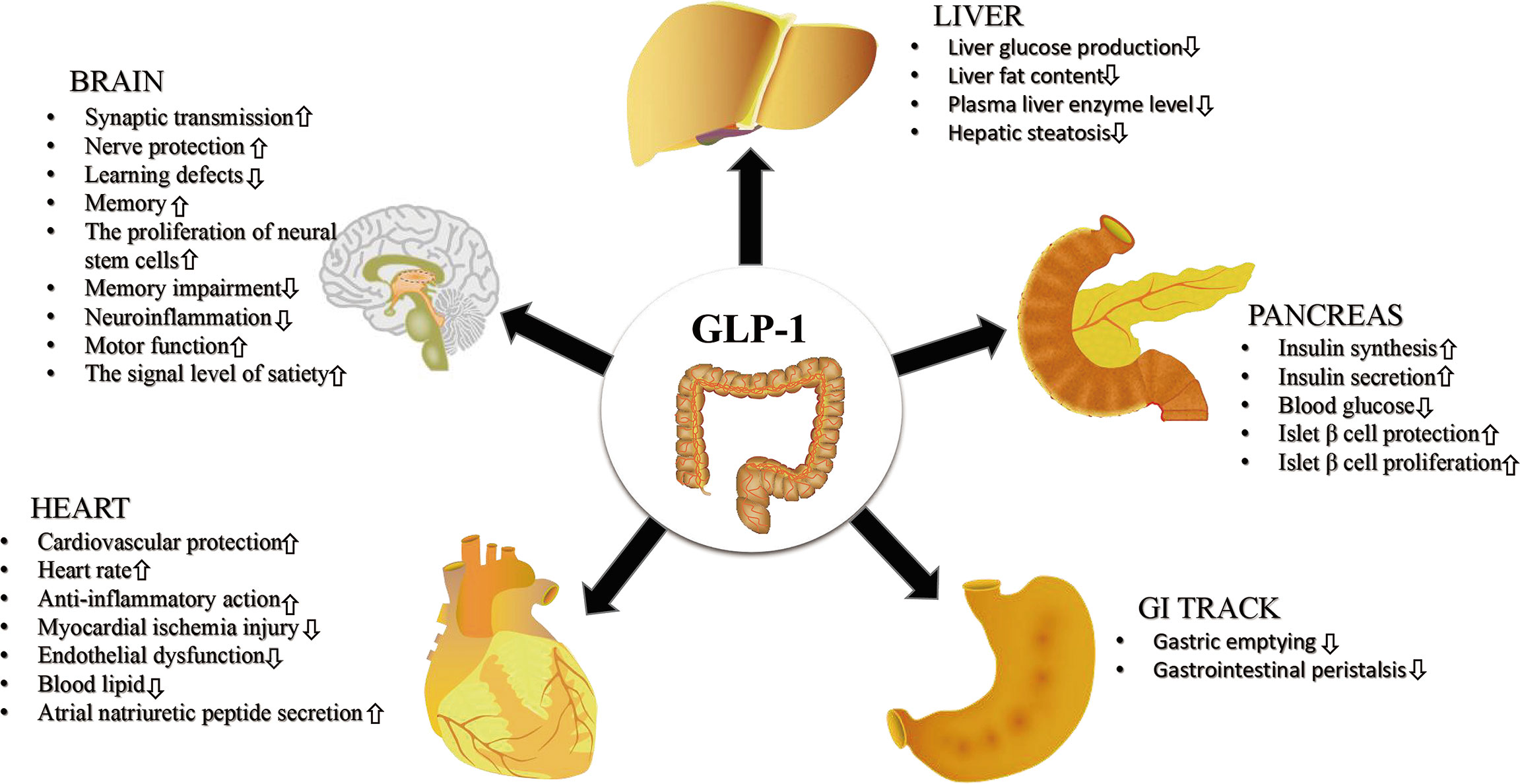
GLP-1 (glucagon-like peptide-1) is famous for its role in glucose control and appetite regulation. But its influence reaches beyond the pancreas. GLP-1 receptors are present in a range of tissues, and downstream signaling can affect vascular function, oxidative stress, and inflammatory activity.
When GLP-1 pathways are activated—whether through incretin-based therapies or related pharmacology—cells may respond with changes in how they handle stress and immune activation. This includes modulation of pathways tied to nuclear signaling and inflammatory gene expression. In plain terms, GLP-1 signaling can influence the conditions under which inflammation either escalates or resolves.
Importantly, inflammation is often “fuelled” by metabolic dysfunction. Insulin resistance, visceral fat accumulation, and dysregulated lipid handling can all contribute to inflammatory tone. By improving metabolic environment, GLP-1–related interventions may indirectly reduce inflammatory burden. Yet there are also hints of direct immunomodulatory effects, suggesting the connection runs deeper than weight changes alone.
Where the Link Comes Together: Shared Pathways and Cellular Cross-Talk
The phrase “link between vitamin D and GLP-1” becomes compelling when you consider shared molecular themes. Both systems interact with immune regulation, oxidative balance, and cell signaling cascades that govern cytokine production. In that sense, vitamin D and GLP-1 may act like two conductors steering the same orchestra—one adjusting the instrument tuning, the other controlling the tempo.
Vitamin D can influence the expression of receptors and regulators involved in immune behavior. GLP-1 signaling can influence inflammatory transcription factors and cellular stress responses. Together, they may shape macrophage polarization and immune cell trafficking—how immune cells choose roles and where they travel. This can alter the tissue microenvironment, potentially affecting both chronic inflammatory diseases and the skin’s tendency toward flare cycles.
A practical way to frame this for readers: vitamin D may reduce “baseline inflammatory readiness,” while GLP-1 signaling may shift “how quickly inflammation escalates” in response to metabolic stress. The combined effect could be synergistic, particularly in individuals who are vitamin D deficient and also experiencing inflammatory-driven metabolic dysregulation.
Inflammation in the Skin: From Barrier Failure to Immune Overdrive
The skin is an inflammatory theater. Keratinocytes, immune cells, and the skin barrier constantly negotiate whether to tolerate or react. When inflammatory signaling becomes dominant, conditions like eczema-like dermatitis, psoriasis, and other inflammatory skin diseases can flare.
Vitamin D’s role here is often discussed in terms of skin differentiation and immune modulation. In immune cells within skin tissue, vitamin D signaling may help restrain exaggerated inflammatory cascades. This can influence redness, scaling, and itch-related pathways—sometimes with effects that reflect improved regulation rather than blunt suppression.
GLP-1–related therapies, while not “skin drugs” in the classic sense, may influence inflammatory tone systemically. Reduced inflammation can translate to fewer flare triggers, better vascular function, and improved oxidative stress handling in skin. Readers may also appreciate the emerging concept of skin–gut–metabolism interplay, where systemic inflammation is shaped by multiple organs rather than living in a single location.

Beyond the Skin: Joints, Gut, and the Inflammatory Web
Inflammation doesn’t keep office hours in one organ. It spreads through signaling networks. In joints, chronic inflammatory processes can drive pain and stiffness, often amplified by immune activity and metabolic factors.
In the gut, immune regulation is even more delicate. Vitamin D can influence the balance between protective and inflammatory immune responses. GLP-1 signaling can affect gut physiology and may modify inflammatory signals through changes in enteroendocrine signaling, barrier integrity, and motility-related mechanisms. The gut, in return, can shape systemic inflammation via metabolites and immune cell cross-talk.
For readers, this section is where “whole-body thinking” becomes essential. Two people can have similar symptoms, yet different underlying causes. Vitamin D status, metabolic health, stress physiology, and baseline immune temperament may determine why one person experiences improvement while another sees minimal change.
What Different Content Types Can Teach Readers
People learn best when information is layered. The topic of vitamin D and GLP-1—two complex systems—benefits from varied content formats. Here’s what readers can realistically expect and how each format supports understanding.
1) Scientific explainers: These break down cytokines, immune cell behavior, receptor signaling, and downstream transcriptional events. They often include diagrams and stepwise logic.
2) Clinical summaries: These translate mechanisms into outcomes: inflammatory markers, symptom scores, and quality-of-life changes. They tend to compare subgroups and discuss time horizons.
3) Practical guides: These focus on daily decisions—vitamin D testing, sun exposure, dietary sources, and how metabolic therapies should be guided by clinicians.
4) Lifestyle narratives: These connect sleep, stress, exercise, and nutrition to inflammatory tone. A long sentence can capture the emotional experience of chronic inflammation; a short one can underline the action step.
5) FAQ-style spot checks: These answer targeted questions such as “Can vitamin D alone calm inflammation?” or “How do GLP-1 therapies relate to immune function?”
6) Case-style storytelling: Fictionalized narratives can show variability—different baselines, different response patterns—without pretending there’s a universal script.
Safety, Testing, and the Importance of Individual Fit
Vitamin D supplementation is not one-size-fits-all. Over-supplementation can be harmful, and deficiency should ideally be confirmed via appropriate testing. The goal is to restore physiological balance, not to chase excessive levels.
Similarly, GLP-1–related therapies require careful medical oversight. They can affect gastrointestinal function and interact with existing health conditions. The best inflammatory outcomes are often tied to correct indications, adherence, and follow-up—rather than improvisation.
Readers should consider a “biological compatibility” mindset: inflammation is influenced by age, body composition, comorbidities, medication history, and baseline vitamin D. In other words, the link between vitamin D and GLP-1 is promising, but it remains personal.
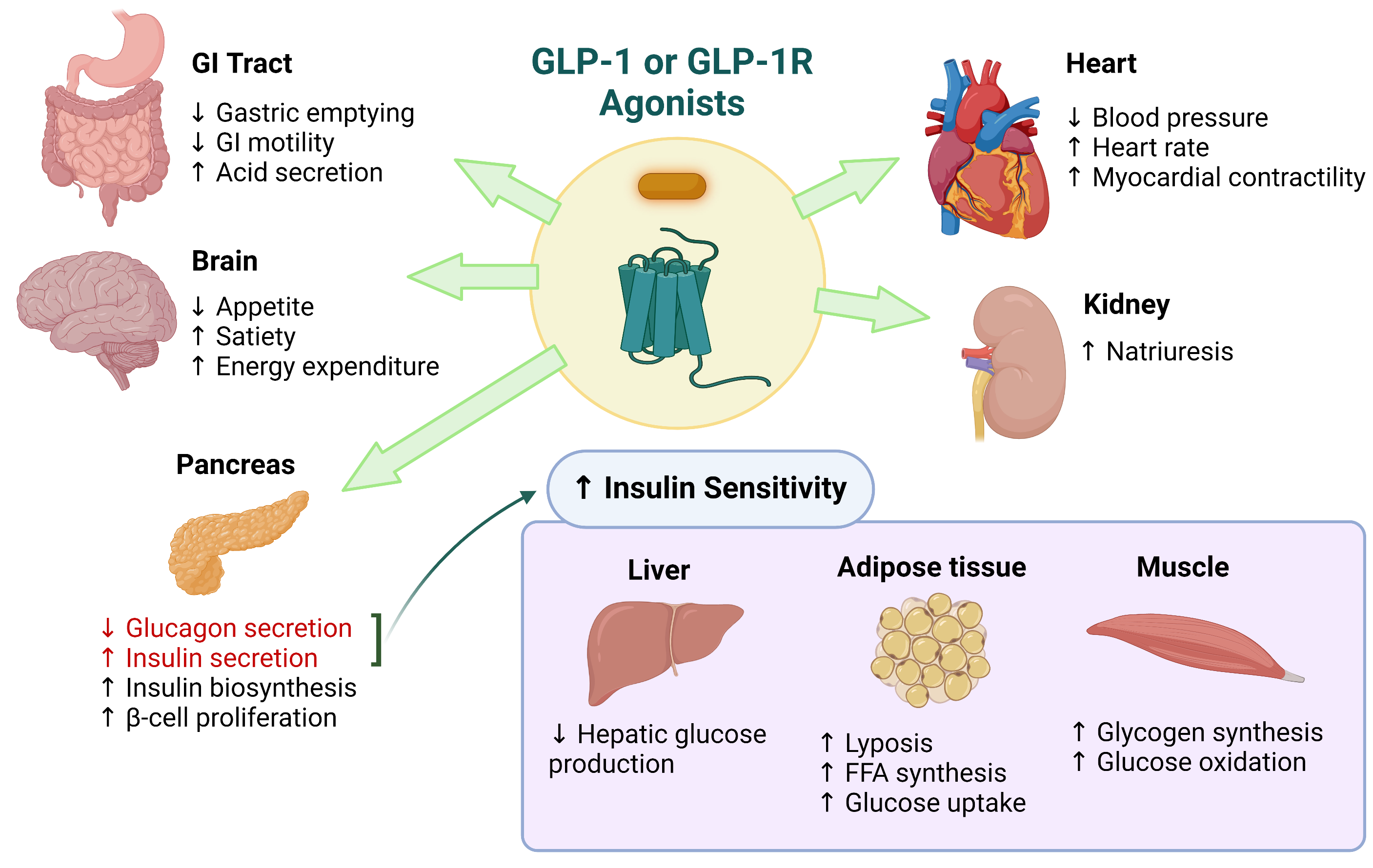
The Takeaway: A Coherent Strategy for Calmer Immunity
The link between vitamin D, GLP-1 signaling, and inflammation reduction is best understood as an alliance of regulatory systems. Vitamin D helps tune immune behavior and reduce inflammatory readiness. GLP-1 pathways can shift metabolic stress, modulate inflammatory signaling, and potentially influence immune tone across tissues. When these effects converge, the body may become less prone to flare-ups and more capable of resolution.
For readers, the most useful approach is not to search for a single miracle mechanism. Instead, imagine a multi-layered plan: confirm vitamin D status, support metabolic health, and allow clinician-guided therapy to target inflammatory drivers. Inflammation is a complex story—but with the right characters in the right roles, the plot can change.