Walk into any pharmacy aisle or scroll through a nutrition forum and the same question tends to rise like a persistent undertow: is 4000 IU of vitamin D truly the upper limit—or is it more myth than measure? The number appears with an air of authority, yet the human body is not a rulebook. It’s a living system where genetics, sunlight exposure, absorption efficiency, kidney function, and even seasonal behavior can radically change how vitamin D behaves. To understand whether 4000 IU is the “true” ceiling, it helps to compare formal regulatory thresholds with the more nuanced interpretations offered by clinicians and researchers.
What “Upper Limit” Really Means in Practice
The phrase “upper limit” can sound like a hard stop, like a governor on an engine. In reality, regulatory limits are usually designed to reduce population-level risk, not to guarantee safety for each individual circumstance. Many agencies define the tolerable upper intake level as the highest daily intake unlikely to cause adverse effects in most people. That definition matters, because it makes the limit statistical rather than absolute.
Vitamin D is fat-soluble, which means it can accumulate. That accumulation is part of why it’s effective—and part of why it can become dangerous when oversupplied. The upper limit exists to limit the probability of hypercalcemia and related complications. Yet hypercalcemia is not triggered by a single number alone. It depends on how much vitamin D is converted into its active forms, how effectively calcium is absorbed, and how strongly the kidneys can excrete excess calcium.
So when readers ask whether 4000 IU is the “true” upper limit, the best response is: it’s a pragmatic ceiling under typical assumptions, but not a universal boundary carved into stone.
How Vitamin D Works: Storage, Activation, and Feedback Loops
Vitamin D isn’t just “a vitamin that goes in.” It’s a sequence of biochemical events. After intake—whether from food, supplements, or sunlight—vitamin D undergoes conversion in the liver into 25-hydroxyvitamin D (25(OH)D), often considered the main circulating indicator of vitamin D status. The kidneys then convert it into the hormonally active form, calcitriol.
Here’s the key nuance: active regulation involves feedback loops. When calcium and calcitriol levels rise, the body attempts to dampen further calcitriol production. However, that regulatory mechanism can be overwhelmed in certain conditions, especially when extremely high intakes persist.
Because vitamin D is stored, short-term spikes can behave differently than long-term supplementation at the same dose. A person taking 4000 IU for a week may experience a very different risk profile than someone taking the same dose for months or years—especially if baseline levels are already high.
Why 4000 IU Became a Common Benchmark
Limits such as 4000 IU often emerge from careful risk modeling. Agencies typically evaluate evidence from studies involving different age groups, evaluate observed adverse effects, and then apply safety factors. The goal is to find a range where most people remain protected, even if they have minor variations in metabolism.
In the public sphere, 4000 IU becomes a symbol. It’s memorable, repeatable, and easy to cite. But a memorable number can still be context-sensitive. Risk hinges on starting vitamin D status. A severely deficient individual may absorb and metabolize differently from someone who already has robust 25(OH)D levels.
In other words, 4000 IU is best understood as a precautionary intake ceiling—especially when taken without laboratory guidance.
FDA-Style Thinking vs Clinical Reality
Regulatory frameworks prioritize broad safety. Clinical practice, meanwhile, is tailored. A clinician might decide that a dose above the everyday upper limit is temporarily appropriate for medically supervised repletion—particularly in cases of proven deficiency, malabsorption syndromes, or certain chronic conditions.
That doesn’t mean “ignore the limit.” It means the limit is not designed to replace clinical judgment. Physicians often emphasize that the risk of toxicity is driven by sustained elevation of 25(OH)D and subsequent effects on calcium homeostasis—not by a single day’s dose.
In regulatory language, the upper limit reduces the likelihood of harm across the general population. In clinical language, the dose becomes a lever: adjusted based on blood tests, symptoms, comorbidities, and follow-up intervals. The same number can represent different realities depending on monitoring.
Expert Opinions: Target Ranges, Not Magic Numbers
Many experts prefer a target-driven approach. Instead of asking only, “Is 4000 IU allowed?” they ask, “What is the current vitamin D status, and what level should be reached safely?” This shifts the conversation from a dose ceiling to a biochemical endpoint.
Clinicians often use serum 25(OH)D to guide dosing, sometimes aiming for levels that reduce deficiency risk while avoiding excessive accumulation. Still, expert opinions vary on what “optimal” means, since different guidelines interpret the evidence differently. Some advocate for modest targets to minimize overtreatment; others accept higher ranges for specific clinical scenarios.
Even among experts, a common thread appears: monitor when doses are high, reassess after a defined period, and pay attention to symptoms that could hint at hypercalcemia (such as unusual thirst, frequent urination, constipation, confusion, or kidney-related discomfort).
When Higher Doses Might Be Considered—Safely
There are situations where higher-than-standard intakes may be used. Typical examples include short-term correction of deficiency, treatment protocols under medical supervision, or cases where absorption is impaired. Malabsorption conditions—such as celiac disease, Crohn’s disease, or after certain bariatric procedures—can dramatically alter vitamin D dynamics.
Another factor is baseline vitamin D status. Someone starting far below normal may require more aggressive repletion. Yet the “more” should be paired with measurement. The most prudent approach often involves trending lab values rather than guessing.
Kidney function also plays a decisive role. If renal conversion or calcium handling is abnormal, the margin for error narrows. Autoimmune conditions and certain granulomatous diseases can also increase risk because extra calcitriol may be produced outside usual regulatory boundaries.
Potential Signs of Excess: What Toxicity Looks Like
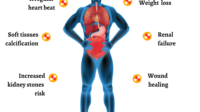
Vitamin D toxicity is less about immediate harm and more about a slow climb toward biochemical imbalance. Excess vitamin D can lead to hypercalcemia, which can strain the kidneys and affect the nervous system, gastrointestinal tract, and cardiovascular system.
Common warning signals can include nausea, vomiting, constipation, dehydration, weakness, and altered mental status. Some people also experience persistent fatigue or bone pain—not because bones “wear out,” but because calcium dysregulation disrupts normal physiology.
Importantly, toxicity is not always obvious. That is why lab monitoring becomes essential when using high doses or when risk factors are present.
How to Choose the Right Strategy: Evidence-Forward and Personalized
A coherent plan often begins with a question: should vitamin D be used at all, and if so, at what dose and for how long? For many readers, a simple strategy includes assessing sunlight exposure, dietary intake, skin pigmentation considerations, latitude, season, body mass index, and age-related changes.
Then come the practical steps. If someone uses moderate supplementation, lab checks might be intermittent. If someone is contemplating doses near or above the 4000 IU threshold—especially long-term—blood testing is typically the safety compass.
Finally, supplementation should be paired with sensible context: adequate calcium intake (not excessive), hydration, and awareness of medications that affect calcium or vitamin D metabolism.
What Types of Content Readers Can Expect Next
To make this topic truly usable, it helps to structure content around different reader needs. Some will want a quick “bottom line,” while others will want a deeper biochemical narrative. Some will benefit from scenario-based guidance; others will prefer a checklist.
Readers can expect discussions that separate myths from mechanisms, outline risk factors for toxicity, and offer clear interpretation of lab trends. Many will also appreciate visual summaries that make dosage ceilings feel less abstract and more actionable.
Below is a simple placeholder-style image that matches the general theme of vitamin-related product imagery—useful for layout or visual storytelling when describing supplement topics.

The Bottom Line: Is 4000 IU “True”?
4000 IU is best viewed as a population-level tolerable upper intake level, designed to minimize risk without laboratory oversight. For most people, exceeding it without monitoring is an unnecessary gamble. Yet the phrase “true upper limit” can mislead—because safety is not merely a number, but a relationship between dose, time, baseline status, and individual physiology.
In clinical reality, doses beyond that benchmark may be used briefly and judiciously under supervision, guided by serum testing and symptom awareness. The most defensible stance is neither panic nor complacency. It’s precision: measure when needed, reassess on schedule, and respect the body’s regulatory limits.