There are treatments that arrive with clinical fanfare, and there are treatments that begin as whispered hypotheses—then, over time, gather momentum in clinics, online communities, and research seminars. The Coimbra Protocol—often described as a high-dose vitamin D approach for multiple sclerosis (MS)—sits firmly in the second category. Advocates describe it as a pragmatic strategy against immune dysregulation; critics warn about safety, study quality, and the seductive simplicity of “one nutrient solves a complex disease.” So is the Coimbra Protocol risky? The most honest answer is: it can be, depending on dosing, monitoring, patient selection, and the quality of medical oversight. To understand why, it helps to unpack what the protocol actually proposes, what evidence exists, and which risks deserve serious attention.
What the Coimbra Protocol Claims to Do
The Coimbra Protocol is widely framed around a bold premise: that vitamin D is not merely a supportive vitamin, but a robust immunomodulator capable of nudulating immune behavior toward less inflammatory activity. Multiple sclerosis involves a cascade of immune-driven processes that damage myelin and disrupt neural signaling. Advocates argue that increasing vitamin D levels helps regulate T-cell activity, influences cytokine patterns, and promotes a more tolerant immune environment.
Rather than focusing on modest supplementation aimed at “reaching sufficiency,” the protocol emphasizes higher dosing intended to elevate circulating 25-hydroxyvitamin D substantially above typical reference ranges. This is where the conversation becomes complicated. Higher levels may produce stronger immunologic effects, but they may also raise the likelihood of adverse metabolic outcomes—particularly in the context of calcium regulation.
Readers should also know that the protocol is not always presented as a single rigid recipe. Some descriptions include additional elements—most notably attention to calcium, magnesium, parathyroid signaling, and overall nutritional context. Others focus mainly on dosing targets. In real-world settings, the protocol’s implementation can vary, which makes consistent safety evaluation difficult.
How High-Dose Vitamin D Is Typically Administered
In most narratives surrounding the Coimbra Protocol, the strategy involves escalating vitamin D doses under clinical supervision, aiming for elevated blood concentrations. The dosing schedule can be dynamic: clinicians may increase or decrease intake depending on lab results and symptom reports.
High-dose vitamin D is often delivered as oral vitamin D3 (cholecalciferol). Because vitamin D is fat-soluble, it can accumulate. That accumulation is not inherently “bad,” but it changes the risk profile. The body must process and store vitamin D effectively, and it must maintain calcium balance—otherwise, the biochemical landscape can turn unfavorable.
What readers may find both reassuring and alarming is this dual nature: a regimen that is conceptually straightforward can become physiologically intricate. Vitamin D doesn’t act in isolation; it interacts with calcium absorption, kidney handling, parathyroid hormone (PTH), and possibly magnesium status.

The Evidence Landscape: Hopeful, Incomplete, and Uneven
The evidence for high-dose vitamin D in MS is often described as promising yet not definitive. Observational studies and smaller trials have suggested associations between higher vitamin D status and reduced MS activity, including lower relapse rates in some groups. Mechanistic research also supports the idea that vitamin D can influence immune pathways relevant to MS.
However, when the Coimbra Protocol is discussed, readers are frequently confronted with a key distinction: correlation does not equal causation. Even when studies show that MS patients with higher vitamin D have better outcomes, it’s not automatic that high-dose supplementation caused the improvement. It’s possible that healthier behaviors, sunlight exposure, or differences in disease severity influenced vitamin D levels.
Additionally, dosage matters. Many studies evaluate “normalization” rather than the comparatively aggressive targets emphasized by high-dose protocols. That makes the safety and efficacy extrapolation less clean. In practice, the evidence gap doesn’t mean the protocol is wrong—it means the medical community still lacks the level of randomized, dose-specific proof that would settle long-term risk questions.
Why the Risk Conversation Centers on Calcium and Kidneys
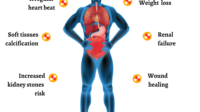
Vitamin D’s strongest physiological downstream effect involves calcium. When vitamin D levels rise significantly, intestinal calcium absorption increases. If calcium becomes too high, the body can experience a cascade of symptoms and complications—ranging from gastrointestinal discomfort to neurologic irritability and, in severe cases, kidney injury through hypercalcemia-related stress.
This is the core reason many experts call high-dose approaches “potentially risky.” The risk is not theoretical; it is biochemically plausible and has known clinical precedents. The kidneys are particularly relevant because they help regulate calcium excretion and overall mineral balance.
Readers should therefore expect that any high-dose protocol worth considering will prioritize monitoring: blood calcium, 25(OH) vitamin D, PTH, and often renal function indicators. Without frequent lab surveillance, the regimen becomes more like an experiment than a therapy.
Potential Side Effects: What Patients Might Notice
Side effects vary depending on dose, baseline health, and timing. When vitamin D is too high relative to the body’s ability to regulate calcium, symptoms of hypercalcemia can emerge. Patients may experience nausea, constipation, fatigue, thirst, frequent urination, confusion, or muscle weakness.
Some adverse effects can be subtle at first. Long sentences of discomfort may begin with short, easily dismissed changes—such as a pattern of dehydration, unusual lethargy, or persistent appetite shifts. This is why clinicians often recommend a structured monitoring plan rather than relying on symptom reporting alone.
It’s also worth noting that MS itself can produce fatigue and sensory changes. That creates a diagnostic blur: symptoms may look like MS progression when they are, in fact, vitamin-related. Proper monitoring helps clarify whether changes are neurological, metabolic, or both.
Who Might Be at Higher Risk and Who Might Be Better Candidates
Not every person with MS faces the same risk profile. Baseline conditions matter. Individuals with a history of kidney stones, chronic kidney disease, sarcoidosis or other granulomatous disorders, or disorders affecting calcium regulation may be more vulnerable to complications from high vitamin D levels.
Medication interactions also deserve attention. Certain drugs can influence calcium balance, renal function, or vitamin D metabolism. Even common prescriptions can change the risk arithmetic.
Additionally, disease state may matter indirectly. A person with active relapses, swallowing or absorption concerns, or inconsistent follow-up may not be an ideal candidate for a high-dose regimen that requires frequent lab checks.
Ultimately, the “risk” question is not a single yes-or-no. It is a probability question tailored to an individual’s physiology, comorbidities, and the rigor of oversight.
Monitoring and Safety Protocol: The Difference Between Care and Recklessness
When a high-dose approach is implemented responsibly, monitoring transforms the entire risk equation. Clinicians should establish targets, timelines, and stop-conditions. They may check 25(OH) vitamin D levels at intervals, evaluate PTH, and assess serum and urine calcium patterns when indicated.
Magnesium status is another frequently overlooked factor. If magnesium is deficient, calcium handling can become erratic, complicating the physiologic story. Dietary calcium intake also matters: excessively high calcium consumption paired with high-dose vitamin D can amplify hypercalcemia risk.
In a well-designed monitoring plan, lab values guide adjustment. Doses can be reduced when vitamin D climbs too high or when PTH signals diminished responsiveness. The safety framework is not merely “measure and hope,” but “measure and calibrate.”

How the Protocol Fits With Standard MS Care
Multiple sclerosis management typically includes disease-modifying therapies, symptom-directed medications, rehabilitation, and lifestyle interventions. The Coimbra Protocol, as discussed in many communities, often appears alongside or instead of conventional care. That is where readers should slow down and consider clinical consequences.
MS relapses can cause cumulative neurological injury. If a high-dose vitamin D approach replaces proven disease-modifying therapy without adequate medical justification, the patient may trade one uncertainty for another. On the other hand, some clinicians view vitamin D as an adjunct—an immunomodulatory support rather than a stand-alone replacement.
A balanced narrative typically treats vitamin D as potentially helpful but not automatically sufficient. The best practice is integration: coordinate with the treating neurologist, maintain evidence-based MS therapy when indicated, and use vitamin D strategy only as a monitored component of a broader plan.
Long-Term Outcomes: The Question That Still Lingers
Long-term outcomes are central to the “risky?” debate. Short-term biochemical improvements do not guarantee durable clinical benefit, and delayed adverse effects may surface only after months or years. High-dose vitamin D is, by design, a sustained exposure strategy—so long-term safety requires long-term data.
Readers should expect ongoing uncertainty, not because the protocol is inherently dubious, but because rigorous dose-specific long-duration trials are difficult. Meanwhile, safety signals—like recurrent hypercalcemia or kidney-related complications—would be expected to appear in the broader clinical record.
Until larger, high-quality studies clarify optimal dosing ranges and monitoring thresholds, risk assessment should remain conservative and individualized.
Content Readers Can Expect: How to Think Critically About Claims
When exploring stories and media about the Coimbra Protocol, readers may encounter several content types. First are patient testimonials and narrative case histories. These can be emotionally compelling and offer context about tolerability and lab changes. They can also be subject to selection bias and cannot establish causality.
Second are mechanistic discussions: explanations of immune modulation, cytokine shifts, and vitamin D receptor effects. These are valuable for understanding plausibility, but plausibility is not the same as proof in diverse patient populations.
Third are video interviews and advocacy-style presentations that emphasize confidence, personal experience, and sometimes unconventional dosing targets. Readers should look for whether the content includes monitoring details, stop conditions, and clear discussion of risks.
Finally, some materials focus on risk management: lab schedules, calcium/PTH interpretation, and contraindication screening. This content is often less sensational but more clinically practical.
The most constructive reading strategy combines empathy with skepticism: appreciate why people seek alternatives, but demand biochemical safeguards and realistic expectations.
So, Is It Risky?
The Coimbra Protocol can be risky when used without rigorous monitoring, inappropriate for certain comorbidities, or when it replaces established MS disease-modifying therapy. The risk is not merely “high-dose equals danger.” Instead, risk arises from the interplay between dose magnitude, individual physiology, and follow-up discipline.
Used responsibly—with individualized assessment, careful lab surveillance, and coordination with neurologic care—it may be less hazardous than critics fear. Used casually or aggressively without supervision, it can become a preventable medical misadventure.
If considering high-dose vitamin D for MS, readers should prioritize one question above all: not just “Does it work?” but “Who is watching the calcium and the kidneys while it tries to work?”